NHS in England poised to deliver boosters
Health officials have sent out marching orders to vaccination clinics.
Vaccination clinics have been given orders to be ready to start delivering boosters jabs “as soon as possible”.
NHS chiefs in England have sent a letter to all local health organisations providing instructions for an imminent start to the booster campaign.
It is expected that the National Bookings System will open on Monday to some people as they become eligible for the jab.
People will be called forward to book six months after they had their second dose.
Most vaccination clinics will begin the booster programme next week but a small number of sites, including some hospitals, could begin sooner.
And care home residents should expect to get their booster by the end of next month.
The letter states that vaccination of health and social care workers “can begin immediately” in some hospitals.
The Joint Committee on Vaccination and Immunisation (JCVI) set out its plan for boosters for more than 30 million British adults on Tuesday.
The JCVI expressed a preference for people to get the Pfizer jab as a third dose, regardless of which jab they were initially given.
But it said that half doses of the Moderna jab could be used as an alternative.
On the co-administration of the flu jab, health leaders said that this should be considered “wherever eligibility for both programmes, supply and regulation allow”.
But the NHS has been keen to stress that people should not delay one vaccine in order to get both jabs at the same time.
GPs have also been ordered to offer the jab to care home residents before November 1.
“Sites should make preparations to start vaccinating as soon as possible,” the letter states.
“It is expected that we will open the National Booking System for bookings and issue first national invitations on Monday September 20.”
The letter adds: “Vaccination of health and social care workers can begin immediately in Hospital Hubs.
“This should be co-administered with flu vaccine wherever possible.”
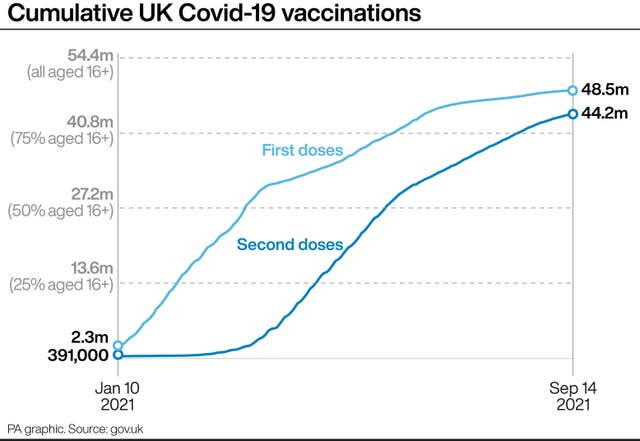
On the vaccine to be used it states: “We do not expect to go live with Moderna half doses immediately.
“Sites should not administer half dose Moderna boosters until instructed to do so.
“In the meantime, systems should begin administering booster vaccinations with Pfizer.”
The news come as an interim analysis on the safety and efficacy of Moderna booster doses was published in the journal Nature.
A small study examined a number of people offered a third dose of the vaccine and variant tweaked booster as part of a phase two trial.
Around 80 participants took part and showed elevated levels of antibodies after a booster.
Commenting on the study, Jonathan Ball, professor of molecular virology at the University of Nottingham, said: “This confirms that an additional boost of an mRNA vaccine, in this case a high or lower dose of the Moderna vaccine, increases the level of virus-killing antibodies in the blood, especially those able to kill variants of concern.
“This is true irrespective of whether the vaccine spike protein sequence is matched to the original strain of virus or the Beta variant.
“Whilst the study stopped short of showing whether it increased protection against infection or disease, it would be reasonable to assume that the higher antibody levels would be beneficial.
“This is one more piece of growing evidence to show that the decision to give boosters to our most vulnerable people over the coming weeks and months was a sensible one.”
Meanwhile, the company behind the Moderna vaccine said its own analysis has shown a lower risk of breakthrough infection – where someone gets the virus even though they have been vaccinated – in those who had their first dose more recently.
The US-based firm said participants in the open-label portion of its phase three study who had their first jab on average eight months previously were at lower risk of contracting coronavirus than those who had their initial jab 13 months previously.
Moderna, which said its study will be submitted for peer-reviewed publication, said its data demonstrates the impact of waning immunity and adds to evidence of the potential benefit of a booster dose.





